According to VSEPR theory the shape of IF 2 – is linear in which iodine is the central atom around which 2 F atoms surround it. how to make a thicc avatar in roblox IF 2 – Lewis Structure Shape. 
It also discusses the bond angle, hybridization, and molecular geometry of IF4+ as well.A step-by-step explanation of how to draw the SbF3 Lewis Dot Structure.For the SbF3 structure use the periodic table to find the total number of valence elec. The …This video shows you how to draw the lewis dot structure for IF4+. The I F 4− has a total number of 36 valence electrons: 7 from iodine, 7 from each of the four fluorine atoms, and 1 from the negative charge. fuse box ford f150 diagram First, start by drawing the Lewis structure for the I F 4− ion. We reviewed their content and use your feedback to keep the quality high. Who are the experts? Experts are tested by Chegg as specialists in their subject area. How many non-bonding electrons are on the central atom in the optimized Lewis structure of OF 2, in which the formal charges are minimized? Expert Answer. For the IF4- structure use the periodic table to find the total number of valence electrons for the IF4- molecule. A step-by-step explanation of how to draw the IF4- Lewis Dot Structure ().The correct number is: 0=0= 0 No, it has the right number of valence electrons but doesn't satisfy the octet rule. O No, it has the wrong number of valence electrons. proposed Lewis structure Is the proposed Lewis structure reasonable? Yes. Decide whether these proposed Lewis structures are reasonable. Assign an AX m E n designation then identify the LP–LP, LP–BP, or BP–BP interactions and predict deviations from ideal bond angles. Determine the electron group arrangement around the central atom that minimizes repulsions. Draw the Lewis electron structure of the molecule or polyatomic ion.Explanation: First, start by drawing the Lewis structure for the I F 4− ion. You examine the molecular and electron-domain geometries of the ion. How can I predict the bond angles for I F 4 −? Chemistry Molecular Orbital Theory Molecular Geometry 1 Answer Stefan V. 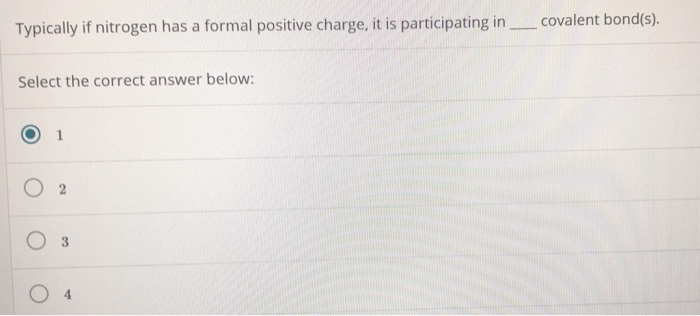
Remember that H \ce IF X 4 X − the skeleton … If there are more than two atoms in the structure, we should place the least electronegative atom as the central atom. Now we will draw a skeleton of given species.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
